History of the International Trichinella Reference Centre (ITRC):
The International
Trichinella Reference Centre (ITRC) was established in 1988 as the official reference laboratory of the International Commission on Trichinellosis (ICT). It was then recognized by the World Organization for Animal Health (OIE) in 1992, and, since 2006, it actively cooperates with the European Union Reference Laboratory for Parasites (EURLP). The ITRC was created as repository of internationally recognized reference strains of
Trichinella and to assist applicants in identifying, at species or genotype level, Trichinella larvae collected from around the world. Over the years, the ITRC has evolved in parallel with the development of new technologies, starting from the early analysis of gene-enzyme systems to the latest molecular techniques. Cheap and fast methods of good sensitivity have been favored. The ITRC assigns to each identified isolate a unique code (ISS code).
The ITRC today: to date, 9491 isolates of human and animal origin from throughout the world have been examined and identified at the species and/or genotype level. These data have been invaluable to establish a taxonomic frame for
Trichinella, which has had a great impact on the epidemiology and clinical management of these zoonotic parasites. More than 20 isolates are maintained
in vivo by periodic passages in CD-1 mice, and the derived reference material (L1 muscle larvae, crude antigens and DNA) is made available for scientists and international institutions involved in studies of
Trichinella biology.
The database: To date, about 70 different host species are recorded in the database, including mammals, birds and reptiles. Among mammals, the largest number of
Trichinella isolates were collected from wild boar (Sus scrofa), domestic pig (Sus scrofa domesticus) and red fox (Vulpes vulpes). Avian hosts are represented by birds of prey or species with scavenging behavior. Reptile species include Nile crocodile (Crocodylus niloticus), saltwater crocodile (Crocodylus porosus) and monitor lizard (Varanus niloticus).
Trichinella isolates collected from pork products (sausages, salami, smoked meat, etc.) and from muscle biopsies of patients with trichinellosis, are scarcely represented.
Scientists from different countries and institutions have contributed to the database growth by providing
Trichinella larvae collected during routine meat inspection of animals intended for human consumption, epidemiological studies, or necroscopy of road-killed or hunted wildlife specimen carcasses. Therefore, since the larvae received by the ITRC were collected for different purposes, the data contained in the database must be interpreted cautiously, taking into account the possible bias due, for example, to epidemiological campaigns on individual host or specific geographic areas.
ITRC users: more than 200 researchers from 52 countries have relied on the ITRC to identify isolates, receive reference material and get epidemiological information and technical support.
Trichinella taxonomy:
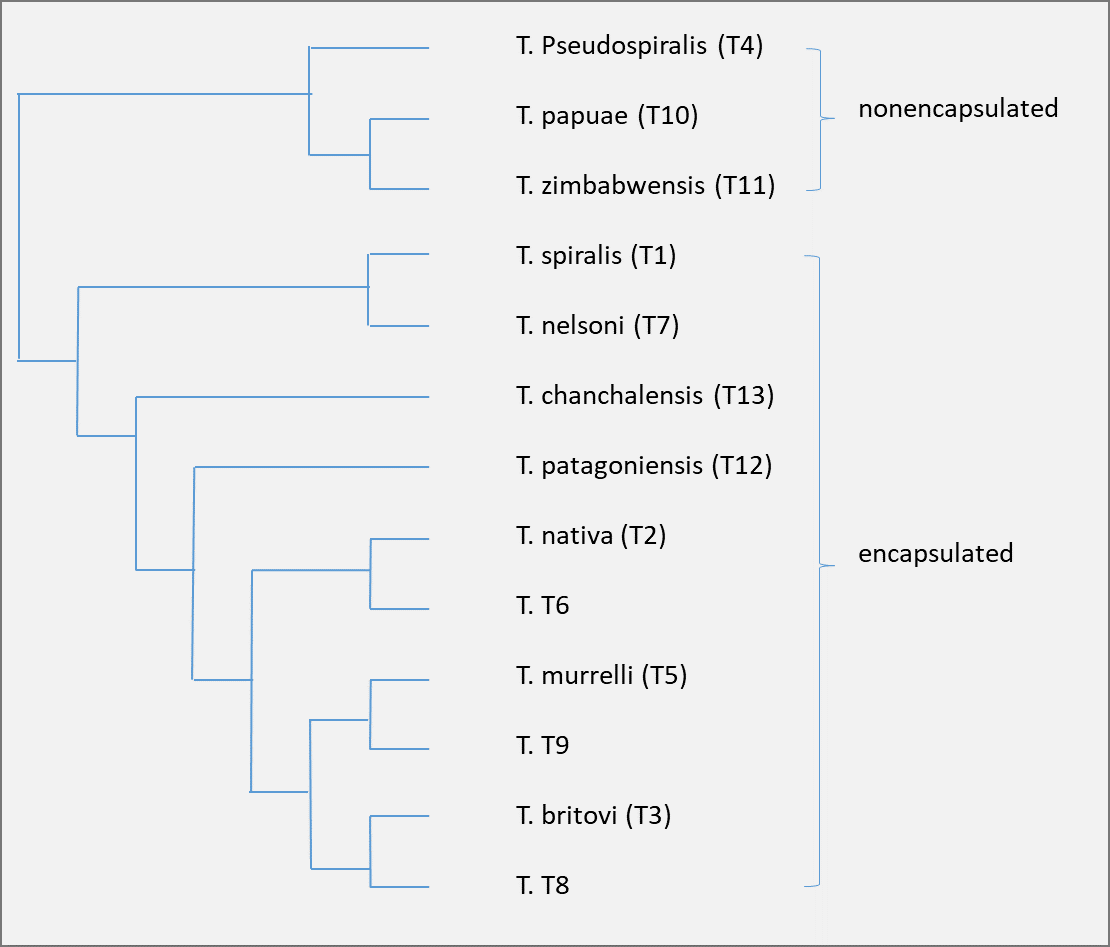
Trichinella systematics and distribution: two main clades are recognized in the genus
Trichinella; one that encompasses nine taxa that encapsulate in host muscle tissue, and a second (including three species) that does not encapsulate following muscle cell differentiation.
Trichinella nematodes show a world-wide distribution in domestic and/or sylvatic animals, with the exception of Antarctica.
The genus
Trichinella belongs to the family Trichinellidae, order Trichocephalida, class Enoplea, and phylum Nematoda. The genus is partitioned into two clades based on the presence or absence of a collagen wall (capsule) that envelopes the worm while in the muscle cell. The encapsulated clade comprises seven species (
T. spiralis, T. nativa, T. britovi, T. murrelli, T. nelsoni, T. patagoniensis and T. chancalensis) and three genotypes (
Trichinella T6,
Trichinella T8 and
Trichinella T9) infecting mammals. The non-encapsulated clade includes three species,
T. pseudospiralis infecting mammals and birds, and
T. papuae and
T. zimbabwensis infecting mammals and reptiles.
Figure 1 - Dendrogram summarizing the hierarchical relationships among taxa (the length of the branches is arbitrary)
 Trichinella
Trichinella life cycle:
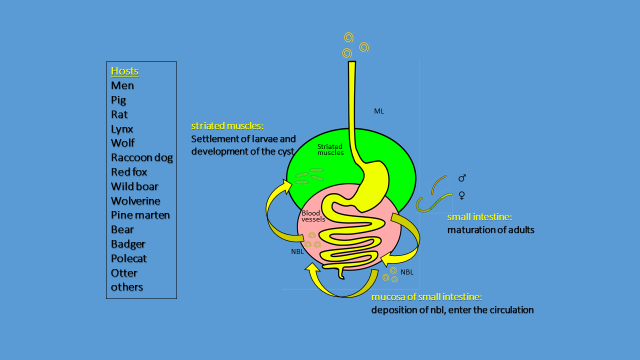
Trichinella life cycle is completed within a single host that harbors all stages, from L1 infective larvae to reproducing adults (i.e. infected animal is both intermediate and definitive host). Infectious L1 larvae reside in the striated muscle and enter the new host when it feeds on infected prey or carcasses. The larvae moult first In the stomach and later in the intestine, and become adults. The ovoviviparous female releases the newborn larvae (NBL) into the intestinal mucosa and these migrate across the entire body of the host through the lymphatic and blood vessels. The larvae that penetrate the striated muscle can settle there and develop into mature L1 larvae. They transform the striated muscle into the nurse cells that provide them with protection and nutrition. Depending on the species, the wall surrounding each larva can be thick and visible (encapsulated species) or so thin that it cannot be observed by light microscopy (non-encapsulated species).
Figure 2 - Schematic
Trichinella spp. life cycle
 The Trichinellosis:
The Trichinellosis:
Trichinellosis is a worldwide zoonosis. Humans acquire the infection by consuming raw or improperly cooked meat or meat-derived products from wild and domestic carnivores and omnivores that are infected with worms of the genus
Trichinella (Figure 1). Domestic animals such as pigs, horses and others, can become infected by contaminated feed or by connection with the wild environment surrounding the farm. At present, human outbreaks due to
Trichinella spp have been reported for most of the species excluding T8,
T. zimbabwensis,
T. patagoniensis and
T. chanchalensis, for which the infectious potential has so far been demonstrated only in laboratory studies on animals.
For more information read the following papers:
- Bruschi, F., 2021. Trichinella and Trichinellosis. Academic Press pp. 1-530.
- Sharma R, Thompson P, Hoberg EP, Scandrett WB, Konecsni K, Harms NJ, Kukka PM, Jung TS, Elkin B, Mulders R, Larter NC, Branigan M, Pongracz J, Wagner B, Kafle P, Lobanov VA, Rosenthal BM, Jenkins EJ, 2020. Hiding in plain sight: discovery and phylogeography of a cryptic species of Trichinella (Nematoda: Trichinellidae) in wolverine (Gulo gulo). Int. J. Parasitol. 50, 277-287.
- Korhonen PK, Pozio E, La Rosa G, Chang BC, Koehler AV, Hoberg EP, Boag PR, Tan P, Jex AR, Hofmann A, Sternberg PW, Young ND, Gasser RB. 2016. Phylogenomic and biogeographic reconstruction of the Trichinella complex. Nat Commun. 7:10513.
- Pozio E. 2015. Trichinella spp. imported with live animals and meat. Vet Parasitol. 213:46-55.
- Pozio E. 2014. Searching for Trichinella: not all pigs are created equal. Trends Parasitol. 30:4-11.
- Pozio E, Zarlenga DS. 2013. New pieces of the Trichinella puzzle. Int J Parasitol. 43:983-97.
- Murrell KD, Pozio E. 2011. Worldwide occurrence and impact of human trichinellosis, 1986-2009. Emerg Infect Dis. 17:2194-202.
- Pozio E. 2007. World distribution of Trichinella spp. infections in animals and humans. Vet Parasitol. 149:3-21.
- Pozio E. 2005. The broad spectrum of Trichinella hosts: from cold- to warm-blooded animals. Vet Parasitol. 132:3-11.
Worldwide occurrence of Trichinella infections: consult the Epidemiological database to know if
Trichinella infection occurs in humans and in domestic and wild animals of a country. The information is based on both old and recent reports published in scientific journals, books, congress abstracts, web sites, etc.